Unit-II: Elimination, Bioavailability and Bioequivalence (10 Hrs.)
Syllabus:
Elimination: Drug metabolism and basic understanding metabolic pathways, renal excretion of drugs, factors affecting renal excretion of drugs, renal clearance, Non renal routes of drug excretion of drugs.
Bioavailability and Bioequivalence: Definition and Objectives of bioavailability, absolute and relative bioavailability, measurement of bioavailability, in-vitro drug dissolution models, in-vitro–in-vivo correlations, bioequivalence studies, methods to enhance the dissolution rates and bioavailability of poorly soluble drugs.
Biotransformation of Drugs/ Metabolism
- The onset of pharmacological response depends upon two pharmacokinetic processes—
- Drug absorption, and
- Drug distribution (since most sites of action are in the extravascular tissues).
- The duration and intensity of action depend upon –
- Tissue redistribution of drug, and
- The rate of drug removal from the body/site of action, i.e. rate of elimination.
- Elimination is the major process for removal of a drug from the body and termination of its action. It is defined as the irreversible loss of drug from the body. Elimination occurs by two processes namely biotransformation and excretion.
- Biotransformation of drugs is defined as the chemical conversion of one form to another. The term is used synonymously with metabolism.
- The chemical changes are usually affected enzymatically in the body and thus, the definition excludes chemical instability of a drug within the body; for e.g. conversion of penicillin to penicilloic acid by the bacterial penicillinase and mammalian enzymes is metabolism but its degradation by the stomach acid to penicillenic acid is chemical instability.
Need for Drug Biotransformation
- All chemical substances that are not nutrients for the body and enter the body through, ingestion, inhalation or absorption are called as xenobiotics(Greek: xenos = foreign) orexogenous compounds.
- Drugs are also xenobiotics which enter the body by virtue of theirlipophilicity. It is interesting to note that for effective absorption, a drug needs to be sufficiently lipid soluble, but it is this same physicochemical property that enables it to bypass excretion.
- This is because only water-soluble agents undergo renal excretion (major route for exit of drugs from the body) whereas lipid soluble substances are passively reabsorbed from the renal tubules into the blood after glomerular filtration. Thus, if such a phenomenon continues, drugs would accumulate in the body and precipitate toxic reactions.
- However, to prevent such a consequence, the body is armed with the metabolic system which transforms the water insoluble, lipophilic, nonpolar drugs into polar and water-soluble products that can be easily excreted by the kidneys and are poorly reabsorbed; for instance, hippuric acid, the metabolite of benzoic acid, is 2.5 times more water-soluble. Drug biotransformation is thus a detoxification process
- Disposition of drug in the body as a consequence of metabolism is illustrates in the Fig. 1.
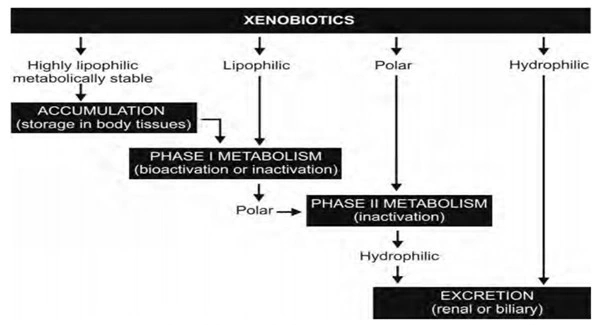
Fig. 1.Disposition of drug in the body as a consequence of metabolism
- Biotransformation –
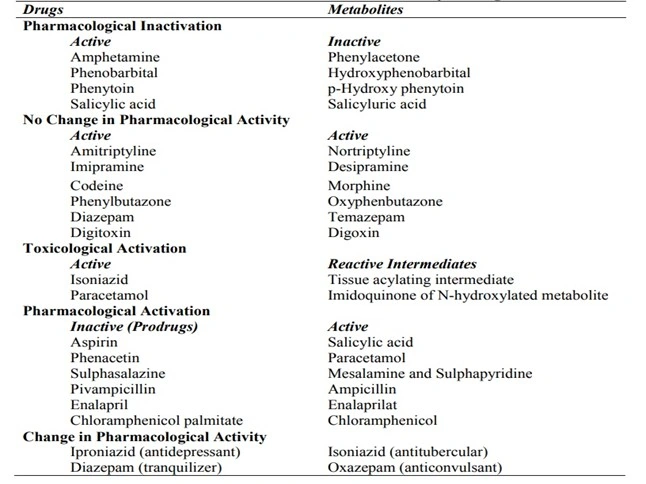
- Normally results in pharmacological inactivation of drugs, i.e. it results in formation of metabolites with little or no pharmacological activity; e.g. conversion of phenytoin to p-hydroxy phenytoin.
- Occasionally yields metabolites with equal activity; e.g. conversion of phenylbutazoneto oxyphenbutazone.
- Rarely leads to toxicological activation of drugs, i.e. it results in formation of metabolites with high tissue reactivity; e.g. conversion of paracetamol to reactive metabolites that cause hepatic necrosis.
- Inactive drugs (prodrugs) also depend upon biotransformation for activation, the process being called as pharmacological activation; A change in pharmacological activity of the drug on metabolism has also been observed.
- TABLE 1. Metabolites and Relative Activity of Drugs
Drug Metabolising Organs
- Liver is the primary site for metabolism of almost all drugs (and other xenobiotics) because of its relative richness in possessing a large variety of enzymes in large amounts. Metabolism by organs other than liver (called as extrahepatic metabolism) is of minor importance sincelower level of drug metabolising enzymes are present in such tissues. The decreasing order of drug metabolising ability of various organs is:
- Liver > Lungs > Kidneys > Intestine > Placenta > Adrenals > Skin. Brain, testes, muscles, spleen, etc. also metabolise drugs but to a small extent.
Drug Metabolising Enzymes
- The enzymes that biotransform xenobiotics differ from those that metabolise food materials. They are versatile and non-specific in metabolising a large number of drugs. The enzymes are broadly divided into 2 categories:
- Microsomal enzymes (within the smooth endoplasmic reticulum)
- Non-microsomal enzymes. (outside the smooth endoplasmic reticulum)
CHEMICAL PATHWAYS OF DRUG BIOTRANSFORMATION
- R.T.Williams, the leading pioneer in drug biotransformation research, divided the pathways ofdrug metabolism reactions into two general categories—
- Phase I reactions, and
- Phase II reactions.
Phase I Reactions
- These reactions generally precede phase II reactions and include oxidative, reductive and hydrolytic reactions. By way of these reactions, a polar functional group is either introduced or unmasked if already present on the otherwise lipid soluble substrate, e.g. -OH (hydroxyl), -COOH (carboxyl), -NH2 (amino) and -SH (sulfhydryl). Thus, phase I reactions are also called as functionalization reactions.
- These transformations are also called as asynthetic reactions, opposite to the synthetic phase II reactions. The resulting product of phase I reaction is susceptible to phase II reactions.
Phase II Reactions
- These reactions generally involve covalent attachment of small polar endogenous molecules such as glucuronic acid, sulphate, glycine, etc. to either unchanged drugs or phase I products having suitable functional groups viz. -OH, -COOH, -NH2 and -SH and form highly water-soluble conjugates which are readily excretable by the kidneys (or bile). Thus, these reactions are called as conjugation reactions.
- Since the outcome of such processes are generally products with increased molecular size, they are also called as synthetic reactions. Quite often, a phase I reaction may not yield a metabolite that is sufficiently hydrophilic or pharmacologically inert but conjugation reactions generally result in products with total loss of pharmacological activity and high polarity. Hence, phase II reactions are better known as true detoxification reactions.
- Since these reactions generally involve transfer of moieties to the substrate to be conjugated, the enzymes responsible are called as transferases.
- The various phase I and phase II reactions are listed in Table 2
TABLE 2. Chemical Pathways of Drug Biotransformation — (M) and (N) Indicate Reactions Catalysed by Microsomal and Non-microsomal Enzymes.
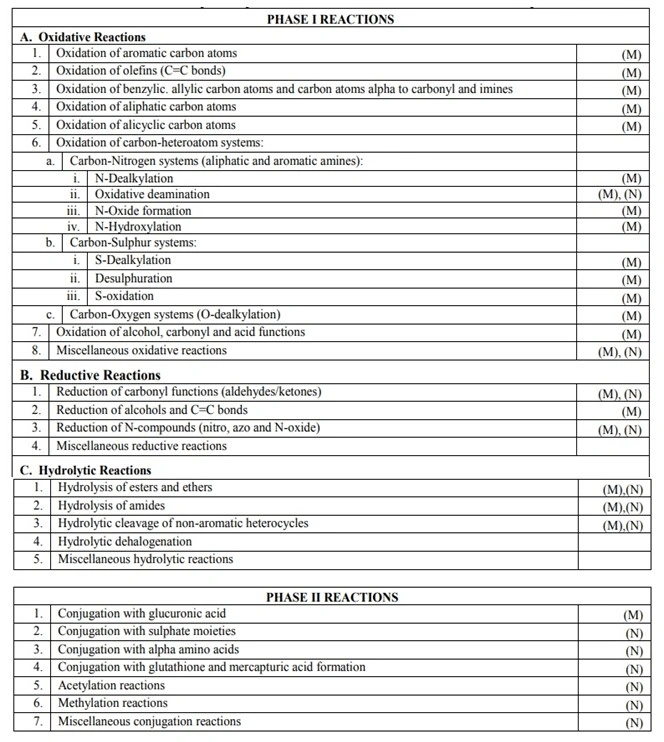
PHASE I REACTIONS
Oxidative Reactions
- Oxidative reactions are the most important and most common metabolic reactions. Almost all drugs that undergo phase I biotransformation undergo oxidation at some stage or the other. A simple reason for oxidation being a predominant reaction is that energy in animals is primarily derived by oxidative combustion of organic molecules containing carbon and hydrogen atoms.
- Oxidative reactions increase hydrophilicity of xenobiotics by introducing polar functional groups such as—OH. Such a polar metabolite can thus rapidly undergo phase II reaction or is excretable by the kidneys.
- Oxidation of xenobiotics is non-specifically catalysed by a number of enzymes located in the microsomes. Such enzymes require both molecular oxygen (O2) and the reducing agent NADPH (Nicotinamide Adenine Dinucleotide Phosphate) to effect reaction. They are therefore referred to as the mixed-function oxidases. The overall stoichiometry of this reaction involving the substrate RH which yields the product ROH, is given by the following equation:
RH + O2 + NADPH + H+ → ROH + H2O + NADP+
Reductive Reactions
- Reductive metabolic reaction can generate polar functional groups such as hydroxy and amino. These groups can undergo further phase I or phase II conjugation reactions. Reduction reaction occurs on various functional groups like aldehyde, ketone, hydroxyl and nitro groups.
Hydrolytic Reactions
- These reactions differ from oxidative and reductive reactions in 3 respects:
- The reaction does not involve change in the state of oxidation of the substrate.
- The reaction results in a large chemical change in the substrate brought about by loss of relatively large fragments of the molecule.
- The hydrolytic enzymes are also involved in metabolism of endogenous substrates and these are found in liver, kidney and intestine.
- A number of functional groups are hydrolysed namely- esters, ethers, amides, hydrazides, etc.
PHASE II REACTIONS
- Phase II reactions involve transfer of a suitable endogenous moiety such as glucuronic acid, sulphate, glycine, etc. in presence of enzyme transferase to drugs or metabolites of phase I reactions having suitable functional groups to form highly polar, readily excretable and pharmacologically inert conjugates.
- Phase II reactions are the real drug detoxication pathways because–
- The conjugates/products of phase II reactions are absolutely free of pharmacological activity.
- The conjugates/products of phase II reactions are highly polar and thus easily excretable either in bile or urine.
- Tissue-reactive and carcinogenic metabolites formed as a result of phase I reaction are rendered harmless by conjugation with moieties such as glutathione.
- The order of capacities of important conjugation reactions is –
Glucuronidation > Amino Acid Conjugation > Sulphation and Glutathione Conjugation
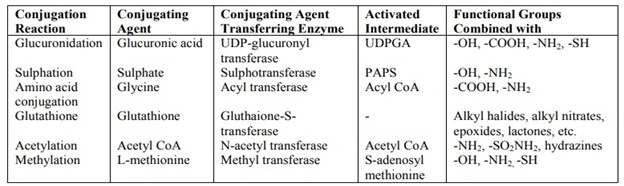
- Table 3 compares the various phase II reactions.
- The molecular weight of the conjugate is important in dictating its route of excretion –
- High molecular weight conjugates (>350) are excreted predominantly in bile
- Low molecular weight conjugates (<250) are excreted in urine.
- Thus, glutathione conjugates are always excreted in bile.
Table 3-Phase II Reactions and their Characteristics
Conjugation With Glucuronic Acid
- Also called as glucuronidation, it is the most common and most important phase II reaction for several reasons:
- Readily available source of conjugating moiety, D-glucuronic acid which is derived from D-glucose.
- Several functional groups viz. alcohols, acids, amines, etc. can combine easily with D-glucuronic acid.
- Quantitatively, conjugation with D-glucuronic acid occurs to a high degree.
- All mammals have the common ability to produce glucuronides,
- The free carboxyl function of glucuronic acid has a pKa in the range 3.5 to 4.0 and hence ionisable at both plasma and urine pH thereby greatly increasing the water solubility of the conjugated substrate.
- The glucuronidation enzymes are in close association with the microsomal mixed function oxidases, the major phase I drug metabolising enzyme system; thus, a rapid conjugation of phase I metabolites is possible.
- Lastly, glucuronidation can take place in most body tissues since the glucuronic acid donor, UDPGA is produced in processes related to glycogen synthesis and thus, will never be deficient unlike those involved in other phase II reactions.
- Glucuronide formation occurs in 2 steps –
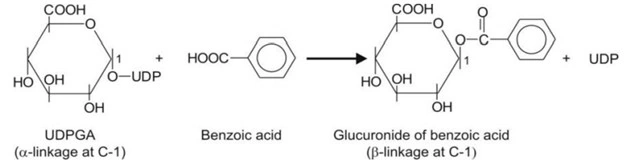
- Synthesis of an activated coenzyme uridine-5′-diphospho-α -D-glucuronic acid(UDPGA) from UDP-glucose (UDPG). The coenzyme UDPGA acts as the donor of glucuronic acid. UDPG is synthesized by interaction of α-D-glucose-1-phosphate with uridine triphosphate (UTP).
- Transfer of the glucuronyl moiety from UDPGA to the substrate RXH in presence of enzyme UDP-glucuronyl transferase to form the conjugate. In this step, the – configuration of glucuronic acid undergoes inversion and thus, the resulting product is – β-D-glucuronide (also called as glucosiduronic acid or glucopyranosiduronic acid conjugate).
The steps involved in glucuronide synthesis are depicted below:

where X = O, COO, NH or S.
An example of glucuronidation of benzoic acid is shown below.
Conjugation With Sulphate Moieties
- Sulphation is similar to glucuronidation but it is catalysed by nonmicrosomal enzymes andoccurs less commonly as the moiety that transfers sulphate to the substrate is easily depleted. This process is thus, easily saturable in comparison to glucuronidation.
- Sulphation is dominant at low substrate concentration, whereas
- Glucuronidation is dominant at high substrate concentration.
- Like glucuronidation, sulphation also occurs in 2 steps:
- Synthesis of an activated coenzyme 3′-phosphoadenosine-5′-phosphosulphate (PAPS)which acts as a donor of sulphate to the substrate.
- Transfer of sulphate group from PAPS to the substrate RXH in presence of enzymesulphotransferase (sulphokinase) and subsequent liberation of 3′-phosphoadenosine-5′-phosphate (PAP).
The steps are summarized in the equations below:

where X = O, NH
Conjugation With Alpha Amino Acids
- This reaction also occurs to a limited extent because of limited availability of amino acids.
- The reaction occurs in two steps:
- Activation of carboxylic acid drug substrate with ATP and coenzyme A (CoA) to forman acyl CoA intermediate. Thus, the reaction is a contrast of glucuronidation and sulphation where the donor coenzyme is activated and not the substrate.
- Acylation of the -amino acid by the acyl CoA in presence of enzyme N-acyltransferase.
The reaction is summarized below.

where R’ = -CH2– (if glycine) or >CH-CH2-CH2-CONH2 (if glutamine)
- Examples of drugs forming glycine or glutamine conjugates are:
- Aliphatic acids e.g. isopropoxyacetic acid
- Alicyclic acids e.g. cholic acid
- Aryl acids e.g. salicylic acid
- Arylacetic acids e.g. phenylacetic acid
- Heterocyclic aryl acids e.g. nicotinic acid.
Conjugation With Glutathione
- Glutathione is a tripeptide with a strongly nucleophilic character due to the presence of a -SH (thiol) group in its structure.
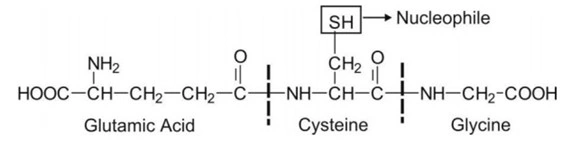
- Thus, it has great affinity for electrophilic substrates, a number of which are potentially toxic compounds. It is important to note that a highly electrophilic metabolite has a tendency to react with tissue nucleophilic groups such as -OH, -NH2 and -SH and precipitate toxicities such as tissue necrosis, carcinogenesis, mutagenesis, teratogenesis, etc. Conjugation with glutathione protects the tissue from such reactive moieties and thus, the reaction is an important detoxication route.
- GSH conjugation differs from other conjugation reactions in that the process does not require initial activation of the coenzyme or the substrate since the GSH, which is anucleophile itself, is highly reactive towards an electrophilic substrate.
- The interaction between the substrate and the GSH is catalysed by enzyme glutathione-S-transferase to form S-substituted glutathione conjugate.
Acetylation
- The general sequence of acetylation reaction is same as that of amino acid conjugation. In acetylation reaction the acetyl group is attached to drug/ metabolite. The enzyme involved is the N-acetyl transferase.
- The drugs/metabolites containing primary amino groups can undergo acetylation. Alcohols (e.g. choline) and thiols (e.g. CoASH) also undergo acetylation with minor extent.
- Acetylation may lead to toxic metabolite, e.g. acetyl derivatives of sulfonamides cause renal toxicity due to reduced water solubility of the metabolites.
Methylation
- It is intermediate reaction between phase I and phase II reactions. The mechanism of methylation reaction is as follow:
- Synthesis of donor of methyl group i.e. activated coenzyme S-Adenosyl Methionine (SAM) from L.-methionine and ATP.
- Transfer of the methyl group from SAM to the drug/metabolite. This reaction is catalyzed by nonmicrosomal enzyme methyl transferase.
- Various methyl transferases responsible for methylation of drug/metabolite are Catechol-O-Methyl Transferase (COMT); Phenyl-O-Methyl Transferase (POMT), Phenyl Ethanolamine-N-Methyl Transferase (PNMT) and nonspecific transferases.
Miscellaneous Conjugation Reactions
Some of the rare conjugation reactions are mentioned below.
Conjugation of Cyanide
Conjugation with Ribose
Conjugation with Taurine
Excretion of Drugs
- Drugs and/or their metabolites are removed from the body by excretion.
- Excretion is defined as the process whereby drugs and/or their metabolites are irreversibly transferred from internal to external environment.
- Excretion of unchanged or intact drug is important in thetermination of its pharmacological action.
- The principal organs of excretion are kidneys. Excretion of drug by kidneys is called as renal excretion. Excretion by organs other than kidneys such as lungs, biliary system, intestine, salivary glands and sweat glands is known as nonrenal excretion.
RENAL EXCRETION OF DRUGS
- Almost all drugs and their metabolites are excreted by the kidneys to some extent or the other. Some drugs such as gentamicin are exclusively eliminated by renal route only.
- Agents that are excreted in urine are –
- Water-soluble.
- Non-volatile.
- Small in molecular size (less than 500 Daltons).
- The ones that are metabolised slowly.
- Nephron: The basic functional unit of kidney involved in excretion is the nephron. Each kidney comprises of one million nephrons. Each nephron is made up of the glomerulus, the proximal tubule, the loop of Henle, the distal tubule and the collecting tubule.
- The principal processes that determine the urinary excretion of a drug are –
- Glomerular filtration.
- Active tubular secretion.
- Active or passive tubular reabsorption.
- These processes are depicted in Fig.3.
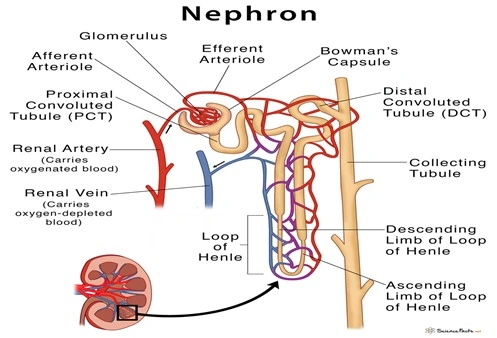
Fig. 2Nephron (information purpose)
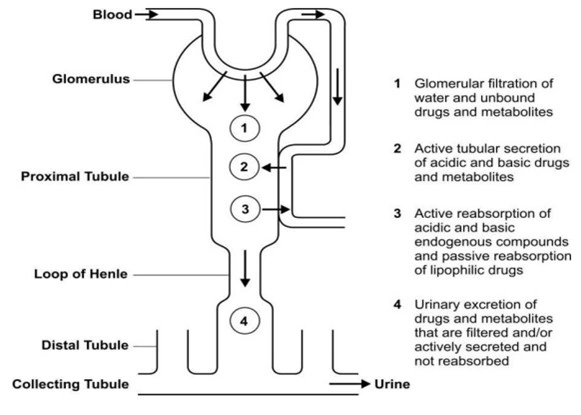
Fig. 3A simplified diagram illustrating processes involved in the urinary excretion ofdrugs
- Glomerular filtration and active tubular secretion tend to increase the concentration of drugs in lumen and hence facilitate excretion whereas tubular reabsorption decreases it and prevents the movement of drug out of the body. Thus, the rate of excretion can be given by equation:
Rate of Excretion = Rate of Filtration + Rate of Secretion – Rate of Reabsorption (Eqn. 1)
Glomerular Filtration
- Glomerular filtration is a non-selective, unidirectional process whereby most compounds, ionised or unionised, are filtered except those that are bound to plasma proteins or blood cells and thus behave as macromolecules.
- The glomerulus also acts as a negatively charged selective barrier promoting retention of anionic compounds. The driving force for filtration through the glomerulus is the hydrostatic pressure of the blood flowing in the capillaries.
- Out of the 25% of cardiac output or 1.2 litres of blood/min that goes to the kidneys via renal artery, only 10% or 120 to 130 ml/min is filtered through the glomeruli, the rate being called as the glomerular filtration rate (GFR).
- Though some 180 litres of protein and cell free ultrafiltrate pass through the glomeruli each day, only about 1.5 litres is excreted as urine, the remainder being reabsorbed from the tubules.
- The GFR can be determined by an agent that is excreted exclusively by filtration and is neither secreted nor reabsorbed in the tubules. The excretion rate value of such an agent is 120 to 130 ml/min. Creatinine, inulin, mannitol and sodium thiosulphate are used to estimate GFR of which the former two are widely used to estimate renal function.
Active Tubular Secretion
- It is a carrier-mediated process which requires energy for transportation of compounds against the concentration gradient.
- The system is capacity-limited and saturable. Two active tubular secretion mechanisms have been identified:
- System for secretion of organic acids/anions like penicillins, salicylates,glucuronides, sulphates, etc. It is the same system by which endogenous acids such as uric acid are secreted.
- System for secretion of organic bases/cations like morphine, mecamylamine,hexamethonium and endogenous amines such as catecholamines, choline, histamine, etc.
- Both the systems are relatively non-selective and independent of each other but both can be bidirectional i.e. agents may both be secreted as well as reabsorbed actively, for example, uric acid.
- Two structurally similar drugs having similar ionic charge and employing the same carrier-mediated process for excretion enter into competition. A drug with greater rate of clearance will retard the excretion of the other drug with which it competes. The half-life of both the drugs is increased since the total sites for active secretion are limited. This may result in accumulation of drugs and thus, precipitation of toxicity. However, the principle of competition can be exploited for therapeutic benefits.
- An interesting example of this is the anionic agent probenecid. Probenecid inhibits the active tubular secretion of penicillins thus increasing their concentration in plasma by at least two fold. A 50% reduction in penicillin G dose is suggested, when it is given with probencid.
Tubular Reabsorption
- Tubular reabsorption occurs after the glomerular filtration of drugs. It takes place all along the renal tubule. Reabsorption of a drug is indicated when the excretion rate values are less than the GFR of 130 ml/min. An agent such as glucose that is completely reabsorbed after filtration has a clearance value of zero. Contrary to tubular secretion, reabsorption results in an increase in the half-life of a drug.
- Tubular reabsorption can either be an:
- Active process, or
- Passive process.
- Active tubular reabsorption is commonly seen with high threshold endogenoussubstances or nutrients that the body needs to conserve such as electrolytes, glucose, vitamins, amino acids, etc. Uric acid is also actively reabsorbed (inhibited by the uricosuric agents). Very few drugs are known to undergo reabsorption actively e.g. oxopurinol.
- Passive tubular reabsorption is common for a large number of exogenous substancesincluding drugs. The driving force for such a process i.e. the concentration gradient is established by the back diffusion or reabsorption of water along with sodium and other inorganic ions.
- Understandably, if a drug is neither secreted nor reabsorbed, its concentration in the urine will be 100 times that of free drug in plasma due to water reabsorption since less than 1% of glomerular filtrate is excreted as urine.
- The primary determinant in the passive reabsorption of drugs is their lipophilicity. Lipophilic substances are extensively reabsorbed while polar molecules are not.
CONCEPT OF CLEARANCE
- The clearance concept was first introduced to describe renal excretion of endogenous compounds in order to measure the kidney function.
- The term is now applied to all organs involved in drug elimination such as liver, lungs, the biliary system, etc. and referred to as hepatic clearance, pulmonary clearance, biliary clearance and so on.
- The sum of individual clearances by all eliminating organs is called as total body clearanceortotal systemicclearance. It is sometimes expressed as a sum of renal clearance and nonrenal clearance.
- Clearance is defined as the theoretical volume of body fluids containing drug from which the drug is removed or cleared completely in a specific period of time. It is expressedin ml/min.
- In comparison to apparent volume of distribution which relates amount of drug in the body to the plasma drug concentration, clearance relates rate of drug elimination to the plasma concentration.

- Renal Clearance (ClR): It can be defined as the volume of blood or plasma which is completely cleared of the unchanged drug by the kidney per unit time. It is expressed mathematically as:

- Physiologically speaking, renal clearance is the ratio of “sum of rate of glomerular filtration and active secretion minus rate of reabsorption” to “plasma drug concentration C”.

FACTORS AFFECTING RENAL EXCRETION OR RENAL CLEARANCE
Factors influencing renal clearance of drugs and metabolites are:
- Physicochemical properties of the drug
- Plasma concentration of the drug
- Distribution and binding characteristics of the drug
- Blood flow to the kidneys
- Biological factors
- Drug interactions
- Disease states
1. Physicochemical Properties of the Drug
- Important physicochemical factors affecting renal excretion of a drug are – molecular size, pKa and lipid solubility.
- The molecular weight of a drug is very critical in its urinary elimination. Drugs of small molecular size with less than 300 Daltons can be easily filtered through the glomerulus and are readily excreted by the kidneys. Drugs in the molecular weight range 300 to 500 Daltons can be excreted both in urine and bile. Molecules of size greater than 500 Daltons are excreted in urine to a lesser extent and excreted through bile.
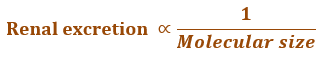
- The influence of drug pKa and lipophilicity on excretion: Urinary excretion of an unchanged drug is inversely related to its lipophilicity. This is because, a lipophilic drug is passively reabsorbed to a large extent.
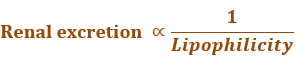
2. Plasma Concentration of the Drug
- Glomerular filtration and reabsorption are directly affected by plasma drug concentration since both are passive processes.
Renal excretion ∝ Plasma concentration of drug
- A drug that is not bound to plasma proteins and excreted by filtration only, shows a linear relationship between rate of excretion and plasma drug concentration.
- In case of drugs which are secreted or reabsorbed actively, the rate process increases with an increase in plasma concentration to a point when saturation of carrier occurs. In case of actively reabsorbed drugs, excretion is negligible at low plasma concentrations. Such agents are excreted in urine only when their concentration in the glomerular filtrate exceeds the active reabsorption capacity, e.g. glucose. With drugs that are actively secreted, the rate of excretion increases with increase in plasma concentration up to a saturation level. These situations are depicted in Fig. 4.
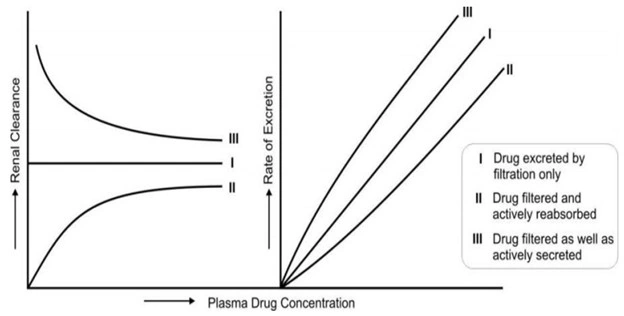
Fig. 4. Renal clearance and rate of excretion of a drug in relation to its plasma concentration as affected by the physiological processes – filtration, active reabsorption and active secretion.
3. Distribution and Binding Characteristics of the Drug
- Clearance is inversely related to apparent volume of distribution of drugs. A drug with large Vd is poorly excreted in urine. Drugs restricted to blood compartment have higher excretion rates.

- Drugs that are bound to plasma proteins behave as macromolecules and thus cannot be filtered through the glomerulus. Only unbound or free drug appear in the glomerular filtrate.
- Drugs extensively bound to proteins have long half-lives because the renal clearance is small and urine flow rate is just 1 to 2 ml/min. The renal clearance of oxytetracycline which is 66% unbound is 99 ml/min while that of doxycycline (7% unbound) is just 16 ml/min.
- Actively secreted drugs are much less affected by protein binding, e.g. penicillins. The free fraction of such drugs are filtered as well as secreted actively and dissociation of drug-protein complex occurs rapidly.
4. Blood Flow to the Kidneys
- The renal blood flow is important in case of drugs excreted by glomerular filtration only and those that are actively secreted. In the latter case, increased perfusion increases the contact of drug with the secretory sites and enhances their elimination.
Renal excretion ∝ Blood flow to kidney
5. Biological Factors
- Age, sex, species and strain differences, differences in the genetic make-up, circadian rhythm, etc. alter drug excretion.
- Renal excretion is approximately 10% lower in females than in males.
- The renal function of newborns is 30 to 40% less in comparison to adults and attains maturity between 2.5 to 5 months of age.
- In old age, the GFR is reduced and tubular function is altered, the excretion of drugs is thus slowed down and half-life is prolonged.
6. Drug Interactions
- Any drug interaction that results in alteration of protein-drug binding characteristics, renal flood flow, active secretion, urine pH and intrinsic clearance and forced diuresis would alter renal clearance of a drug.
- Alteration of Urine pH: Acidification of urine with ammonium chloride, methionineor ascorbic acid enhances excretion of basic drugs.
Alkalinisation of urine with citrates, tartarates, bicarbonates and carbonic anhydrase inhibitors enhances excretion of acidic drugs. - Competition for Active Secretion: the drug which utilize the same carrier for active tubular secretion, can compete with each other for binding with carrier which affects excretion.
- Forced Diuresis: All diuretics increase elimination of drugs whose renal clearancegets affected by urine flow rate.
7. Disease States—Renal Impairment
- Renal dysfunction greatly impairs the elimination of drugs especially those that areprimarily excreted by the kidneys. Some of the causes of renal failure are hypertension, diabetes mellitus, hypovolemia (decreased blood supply to the kidneys), pyelonephritis (inflammation of kidney due to infections, etc.), nephroallergens (e.g. nephrotoxic serum) and nephrotoxic agents such as aminoglycosides, phenacetin and heavy metals such as lead and mercury.
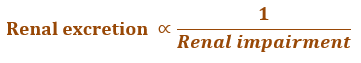
- Uraemia, characterized by impaired glomerular filtration and accumulation of fluids andprotein metabolites, also impairs renal clearance of drugs. In both these conditions, the half-lives of drugs are increased. As a consequence, drug accumulation and toxicity may result. Determination of renal function is therefore important in such conditions in order to monitor the dosage regimen.
NON-RENAL ROUTES OF DRUG EXCRETION
Drugs and their metabolites may also be excreted by routes other than the renal route, called as the extrarenal ornonrenal routes of drug excretion. The various such excretionprocesses are:
- Biliary excretion (Liver-Faeces)
- Pulmonary excretion (Lungs)
- Salivary excretion
- Mammary excretion (Breast milk)
- Skin/dermal excretion
- Gastrointestinal excretion
- Genital excretion (Reproductive organ)
1. Biliary Excretion of Drugs
- Bile is produced by the hepatic cells lining the bile canaliculi.
- It is produced in liver and transported from the liver through the hepatic ducts, stored and concentrated in the gallbladder, and released into the duodenum (the first part of the small intestine) via the common bile duct, stimulated by meals.
- In humans, the bile flow rate is steady 0.5 to 1 mL/min. Almost 90% of the secreted bile acids are reabsorbed from the intestine and transported back to the liver for re-secretion. The rest is excreted in faeces.
- A drug, whose biliary concentration is less than that in plasma, has a small biliary clearance and vice versa.
- A higher bile/plasma concentration ratio generally indicates more excretion of a compound into the bile.
- Compounds that are excreted in bile have been classified into 3 categories on the basis of their bile/plasma concentration ratios:
- Group A compounds whose ratio is approximately 1, e.g. sodium, potassium and chloride ions and glucose.
- Group B compounds whose ratio is >1, usually from 10 to 1000, e.g. bile salts, bilirubinglucuronide, creatinine, sulphobromophthalein conjugates, etc.
- Group C compounds with ratio < 1, e.g. sucrose, inulin, phosphates, phospholipids andmucoproteins.
- Several factors influence secretion of drugs in bile –
- The most important factor governing the excretion of drugs in bile is their molecular weight. Its influence on biliary excretion is summarized in the below Table.
- Polarity is the other physicochemical property of drug influencing biliary excretion. Greater the polarity, better the excretion. Thus, metabolites are more excreted in bile than the parent drugs because of their increased polarity.

- Orally administered drugs which during absorption process go to the liver, are excreted more in bile in comparison to parenterally administered drugs.
- The ability of liver to excrete the drug in the bile is expressed by biliary clearance.

2. Pulmonary Excretion
- Pulmonary excretion is the process by which drugs, specifically gaseous and volatile substances, are eliminated from the body through exhaled air.
- Gaseous and volatile substances such as the general anaesthetics (e.g. halothane) are absorbed through the lungs by simple diffusion. Similarly, their excretion by diffusion into the expired air is possible.
- Factors influencing pulmonary excretion of a drug include pulmonary blood flow, rate of respiration, solubility of the volatile substance, etc.
- Cardiac Output: High blood flow to the lungs increases the amount of drug delivered for excretion.
- Respiration Rate: Faster breathing speeds up the removal of the drug from the alveolar space, maintaining the concentration gradient.
- Compounds like alcohol which have high solubility in blood and tissues are excreted slowly by the lungs.
3. Salivary Excretion
- Salivary excretion is a minor route of drug elimination where substances pass from the blood plasma into the saliva.
- Most drugs enter saliva through passive diffusion across the epithelial cells of the salivary glands.
- Selective Entry: Only the unbound, lipid-soluble, and unionized form of a drug can easily cross these membranes.
- Active Transport: Some drugs, such as metformin and penicillin are transported into saliva via specialized proteins like the organic cation transporter OCT3.
- Recycling: Because saliva is typically swallowed, many excreted drugs are reabsorbed in the gastrointestinal tract, leading to a form of internal recycling rather than permanent elimination from the body.
- Basic drugs are excreted more in saliva as compared to acidic drugs.
- Drug excreted in saliva, e.g. Penicillin, caffeine, theophylline, phenytoin, carbamazepine, etc.
- The bitter after taste in the mouth of a patient on medication is an indication of drug excretion in Saliva.
4. Mammary Excretion (Breast milk)
- Mammary excretion is the process by which drugs and their metabolites pass from a mother’s systemic circulation into breast milk. While it is a minor route for the mother’s elimination.
- Excretion of a drug in milk is clinically critical/ important since it can gain entry into the breast-feeding infant.
- Drugs enter breast milk primarily through passive diffusion across the epithelial membrane of the mammary glands.
- The “Blood-Milk Barrier”: During the first few days after birth (the colostrum phase), the gaps between mammary cells are large, allowing almost all drugs to pass easily. As the milk matures, these gaps close, making the barrier more selective.
- Lipid Solubility: Since breast milk has a higher fat content than plasma, lipid-soluble (lipophilic) drugs (like diazepam) concentrate more easily in milk.
- Since milk is acidic (6.6 to 7.4) in comparison to plasma (7.0 to 7.2), weakly basic drugs concentrate more in milk.
- Drugs extensively bound to plasma proteins, e.g. diazepam, are less secreted in milk.
- The amount of drug excreted in milk is generally less than 1% and the fraction consumed by the infant is too less to reach therapeutic or toxic levels. But some potent drugs such as barbiturates, morphine and ergotamine may induce toxicity in infants. Some examples of toxicity to breast-fed infants owing to excretion of drug in milk are –
- Chloramphenicol: Possible bone marrow suppression.
- Diazepam: Accumulation and sedation.
- Heroin: Prolonged neonatal dependence.
- Methadone: Possible withdrawal syndrome if breast-feeding is stopped suddenly.
- Propylthiouracil: Suppression of thyroid function.
- Tetracycline: Permanent staining of infant teeth.
- Wherever possible, nursing mothers should avoid drugs. If medication is unavoidable, the infant should be bottle-fed.
5. Skin Excretion
- Skin excretion is a minor route of drug elimination where substances are removed from the body through sweat or sebum.
- Drugs reach the skin surface through two main glands:
- Eccrine Sweat Glands: Most drugs enter sweat via passive diffusion from the blood capillaries surrounding the glands. Only the unbound (free) and unionized form of a drug typically crosses into sweat.
- Sebaceous Glands: These glands secrete sebum (skin oil). Highly lipid-soluble (lipophilic) drugs tend to accumulate here, as sebum is rich in fats.
- Factors Influencing Skin Excretion
- pH Partitioning: Sweat is typically more acidic (pH ~4.0 to 6.8) than blood (pH 7.4). This can cause basic drugs (like amphetamines) to become “trapped” in sweat at higher concentrations than in the blood.
- Lipid Solubility: Since the skin surface and sebum are fatty, drugs with high lipid solubility (like griseofulvin or ketoconazole) are excreted more efficiently this way.
- Sweat Rate: Physical activity, high ambient temperature, or certain fevers increase sweat production, which can accelerate the excretion of water-soluble drugs like urea or heavy metals.
6. Gastrointestinal Excretion
- Excretion of drugs into the GIT usually occurs after parenteral administration when the concentration gradient for passive diffusion is favourable. The process is reverse of GI absorption of drugs.
- Water soluble and ionised form of weakly acidic and basic drugs is excreted in the GIT, e.g. nicotine and quinine are excreted in to stomach. Orally administered drugs can also be absorbed and excreted in the GIT.
- Drugs excreted in the GIT are reabsorbed into the systemic circulation and undergo recycling.
7. Genital Excretion
- Genital excretion refers to the elimination of drugs or their metabolites through the secretions of the male or female reproductive tracts, specifically semen (seminal fluid) and vaginal secretions.
- Drugs enter genital secretions primarily via passive diffusion from the blood plasma across the epithelial barriers of the prostate, seminal vesicles, or vaginal wall.
- The Blood-Testis/Blood-Prostate Barrier: These are tight junctions that limit the passage of many drugs. Only lipid-soluble, unbound, and unionized drugs can easily cross.
- Vaginal fluid is also acidic (pH ~4.0–4.5), which can similarly influence the concentration of basic drugs.
A summary of drugs excreted by various routes is given in below Table.
| Excretory Route | Mechanism | Characteristics of Drugs Excreted |
| Renal excretion | Glomerular filtration, active secretion, active/passive reabsorption | Free, hydrophilic, unchanged drugs/metabolites/conjugates with Molecular Weight (MW) < 500. |
| Biliary excretion (Liver-Faeces) | Active secretion | Hydrophilic, unchanged drugs/metabolites/conjugates with MW ≥ 500. |
| Pulmonary excretion (Lungs) | Passive diffusion | Gaseous and volatile substances; drugs that are insoluble in blood and tissue. |
| Salivary excretion | Passive diffusion, active transport | Free, unionised, lipophilic drugs; some polar drugs. |
| Mammary excretion (Breast milk) | Passive diffusion | Free, unionised, lipophilic drugs (generally basic in nature). |
| Skin/dermal excretion (Sweat) | Passive diffusion | Free, unionised, lipophilic drugs. |
| Gastrointestinal excretion | Passive diffusion | Water-soluble, ionised drugs. |
| Genital excretion (Reproductive organ) | Passive diffusion | lipid-soluble, unbound, basic (vegina) and unionized drugs |
Bioavailability and Bioequivalence
- Bioavailability is defined as the rate and extent (amount) of absorption of unchanged drug from its dosage form into the systemic circulation.
- Bioequivalence is a comparative term. Two drug products are considered bioequivalent if their bioavailabilities do not show a significant difference when administered at the same dose under similar conditions.
Bioavailability:
- The therapeutic effectiveness of a drug depends upon the ability of the dosage form to deliver the medicament to its site of action at a rate and amount sufficient to elicit the desired pharmacological response. This attribute of the dosage form is referred to as physiological availability, biological availability or simply, bioavailability.
- For mostdrugs, the pharmacological response can be related directly to the plasma levels. Thus, the term bioavailability is defined as the rate and extent (amount) of absorption of unchanged drug from its dosage form into the systemic circulation.
- The rate or rapidity withwhich a drug is absorbed is an important consideration when a rapid onset of action is desired as in the treatment of acute conditions such as asthma attack, pain, etc. A slower absorption rate is, however, desired when the aim is to prolong the duration of action or to avoid the adverse effects. On the other hand, extent of absorption is of special significance in the treatment of chronic conditions like hypertension, epilepsy, etc.
- If the size of the dose to be administered is same, then bioavailability of a drug from its dosage form depends upon 3 major factors:
- Pharmaceutical factors related to physicochemical properties of the drug andcharacteristics of the dosage form.
- Patient-related factors.
- Route of administration.
- The former two factors (pharmaceutical factors and patient-related factors) have been dealt with comprehensively in chapter–Absorption of Drugs.
- The influence of route of administration on drug’s bioavailability isgenerally in the following order with few exceptions:
Parenteral > Oral > Rectal > Topical
- Within the parenteral route, intravenous injection of a drug results in 100% bioavailability as the absorption process is bypassed. However, for reasons of stability and convenience, most drugs are administered orally. In such cases, the dose available to the patient, called as the bioavailable dose, is often less than the administered dose.
- The amount of drug that reaches the systemic circulation (i.e. extent of absorption) is called as systemic availabilityor simplyavailability.
- The term bioavailable fraction F, refers to the fraction of administered dose that enters the systemic circulation.

Objectives of Bioavailability Studies
Bioavailability studies are important in the —
- Primary stages of development of a suitable dosage form for a new drug entity to obtain evidence of its therapeutic utility.
- Development of new formulations of the existing drugs.
- Determination of influence of excipients, patient related factors and possible interaction with other drugs on the efficiency of absorption.
- Control of quality of a drug product during the early stages of marketing in order to determine the influence of processing factors, storage and stability on drug absorption.
- Comparison of availability of a drug substance from different dosage forms or from the same dosage form produced by different manufacturers.
